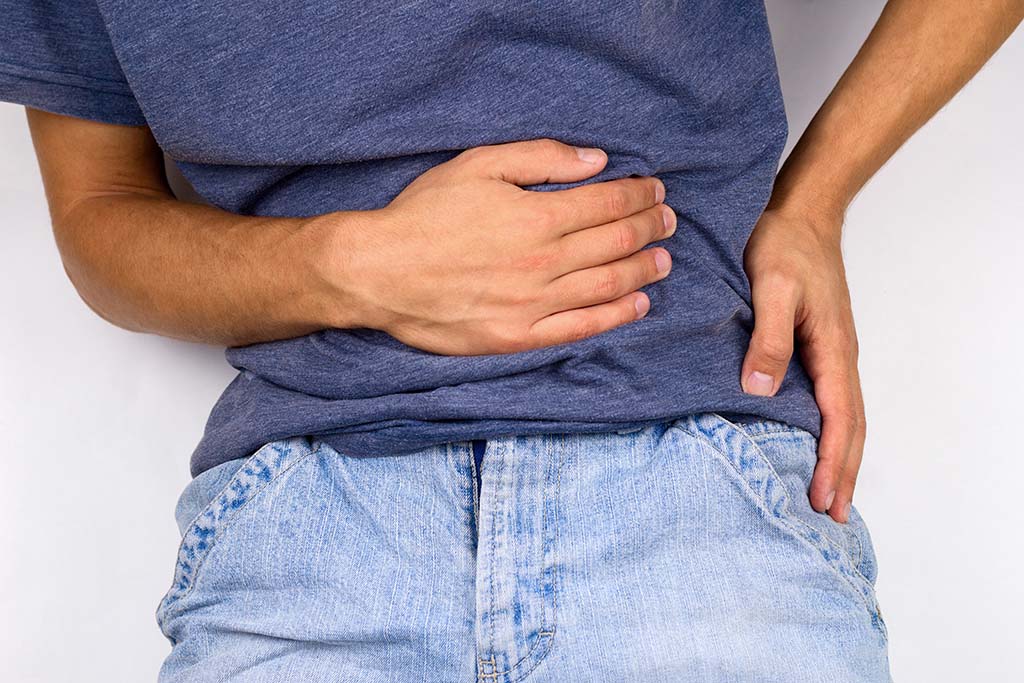
Abdominal mesh, a medical device that has been widely used to treat hernias, has been the subject of thousands of lawsuits filed by patients who experienced sometimes significant side effects. The mesh is implanted in the abdomen, groin, or stomach to support tissue that has been damaged – usually by a hernia – and the most common adverse effects include infection, abdominal pain, bowel obstruction, pain, adhesion, and hernia recurrence.
One hernia mesh product was removed without being recalled, and four were recalled from the market after they were shown to have led to serious complications. The four that were recalled are: Genitrix Surgical Matrix, Atrium Medical C_QUR Mesh, Bard Composix Kugel Hernia Patch, and Ethicon Proceed Surgical Mesh. However, products that have not been recalled or removed from the market have also been found to have caused damages.
Filing a successful case requires submitting the correct documentation, including the mesh implant manufacturer, the date of surgery, details relating to the medical appointment, and all medical documentation related to the case. Once you have that documentation available, it is highly important that you work with an attorney with experience handling abdominal mesh cases. Most well-documented cases should result in damages being awarded, but a failure to provide the correct documentation may delay or derail the lawsuit.
To contact the experienced hernia mesh lawyers at Napoli Shkolnik, click here. To read a more detailed blog post relating to hernia mesh cases, click here.